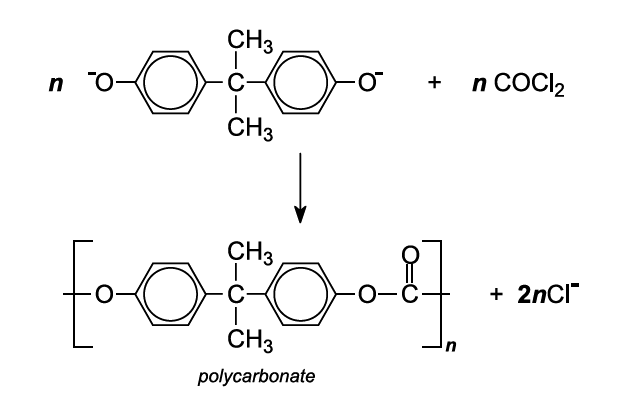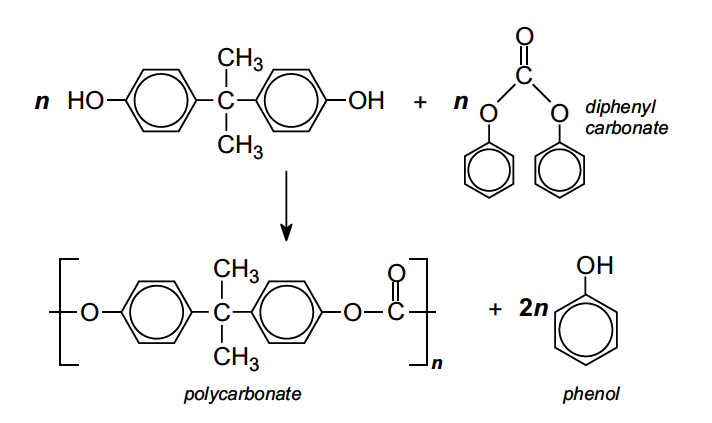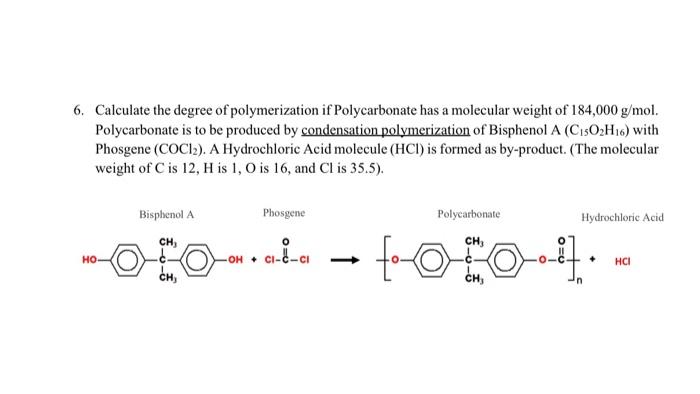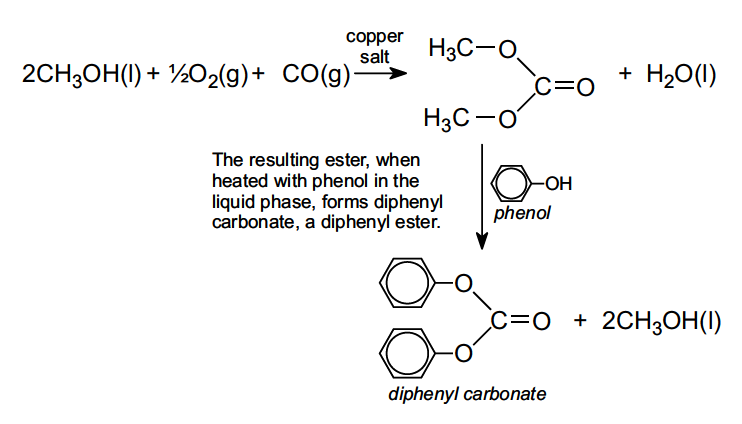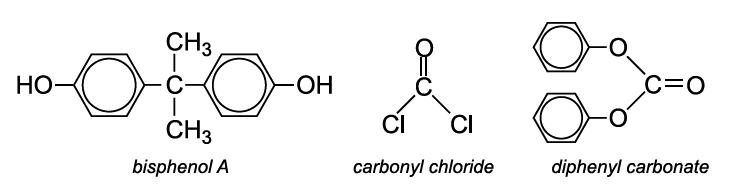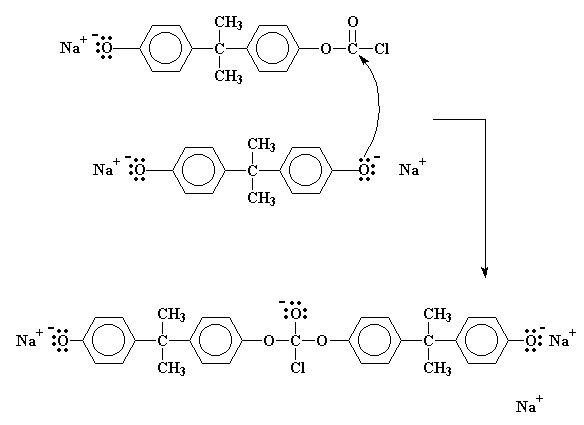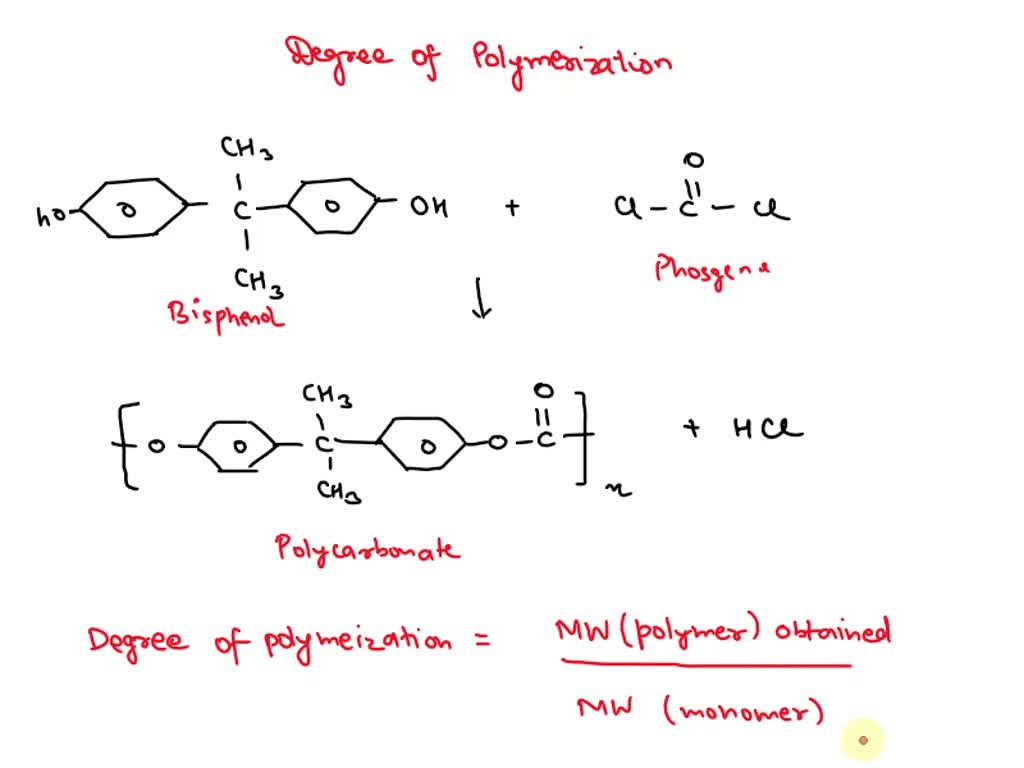
SOLVED: Calculate the degree of polymerization if Polycarbonate has a molecular weight of 184,000 g/mol. Polycarbonate is to be produced by condensation polymerization of Bisphenol A (C15H16O2) with Phosgene (COCl2). A Hydrochloric
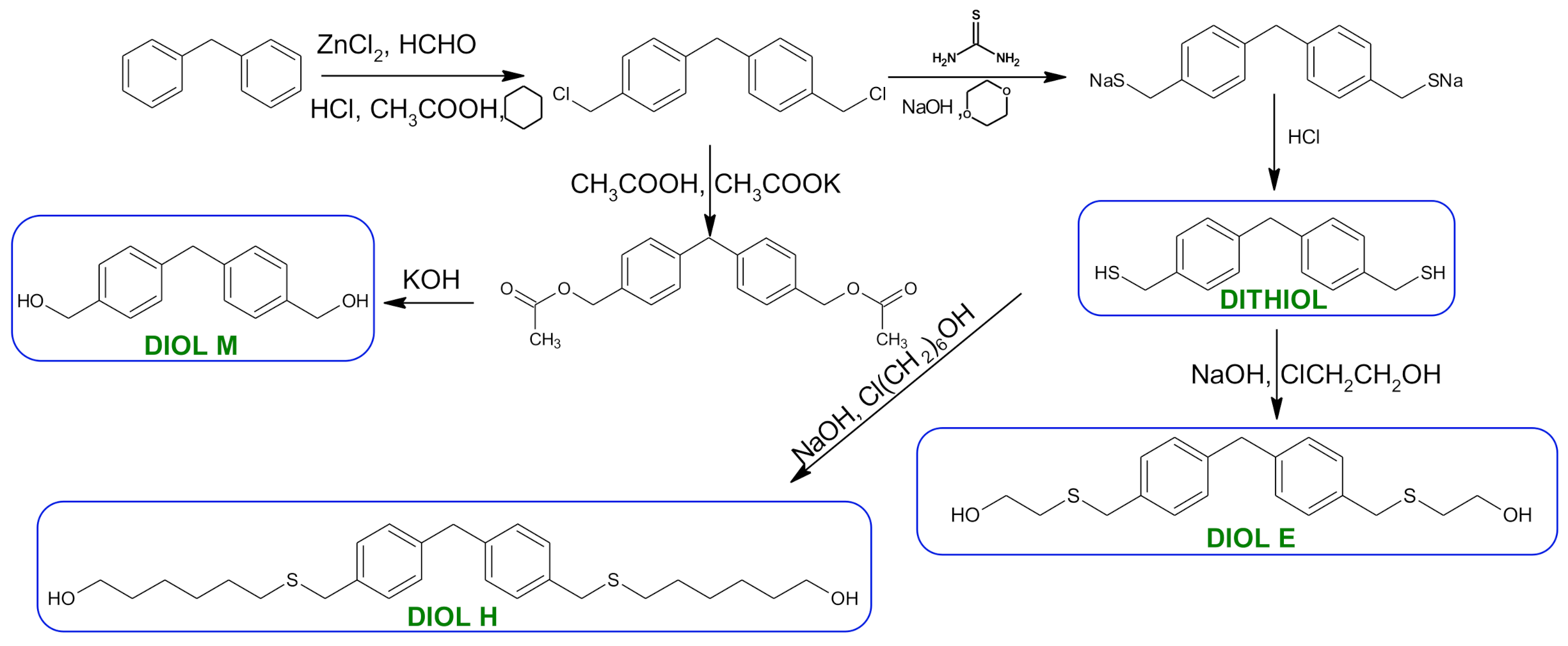
Polymers | Free Full-Text | Synthesis and Spectroscopic Analyses of New Polycarbonates Based on Bisphenol A-Free Components
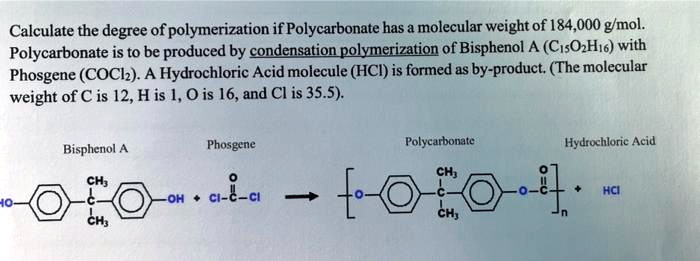
SOLVED: Calculate the degree of polymerization if Polycarbonate has a molecular weight of 184,000 g/mol. Polycarbonate is to be produced by condensation polymerization of Bisphenol A (C15H16O2) with Phosgene (COCl2). A Hydrochloric